|
4/9/2024 0 Comments Democritus atomic theory 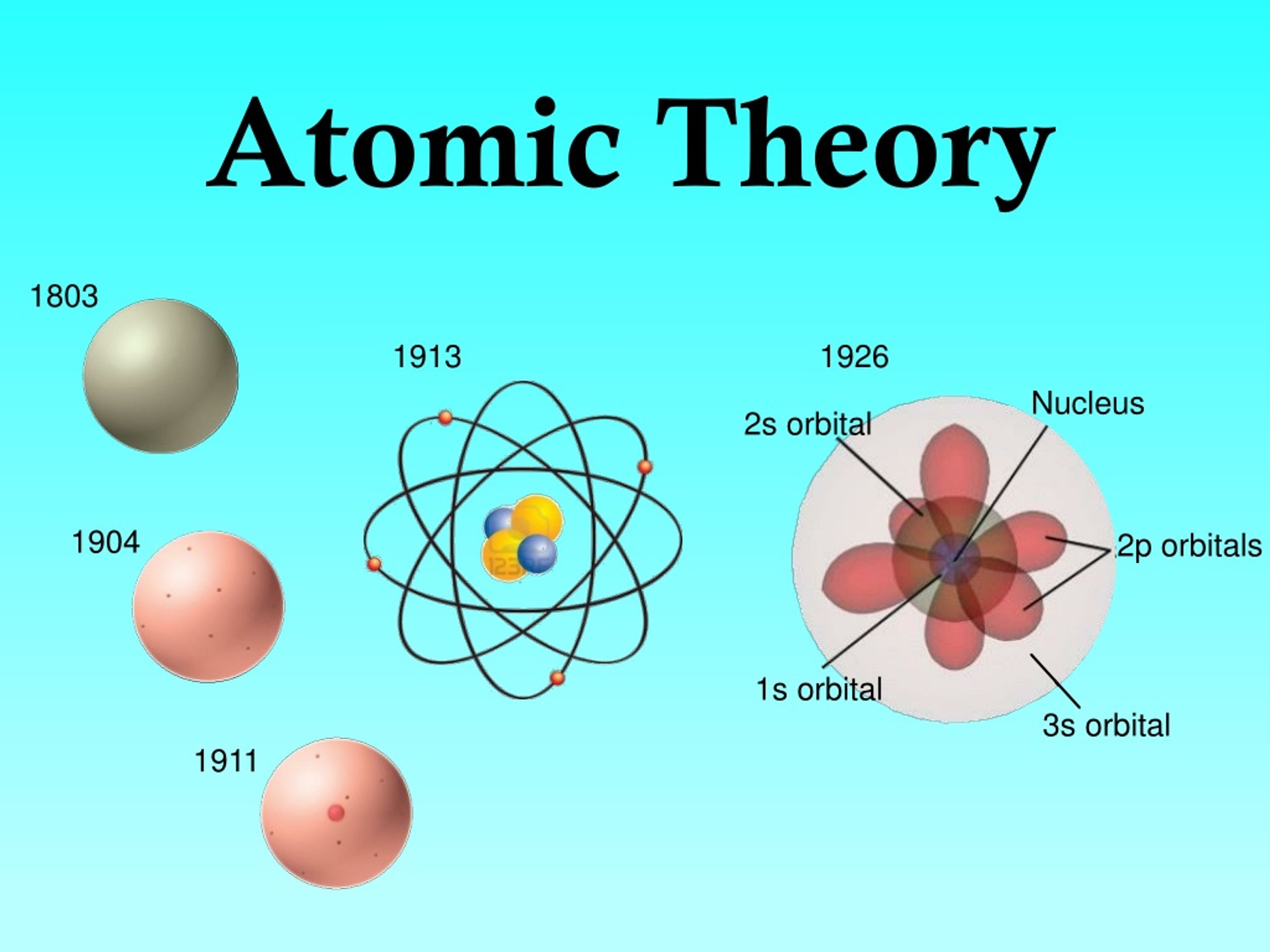
This idea is an extension of a law published in the late 1700s by Joseph Proust. For example, the compound water (H 2O) is always a combination of one oxygen atom and two hydrogen atoms. What is it really saying? It's saying that compounds are collections of atoms put together in very specific whole number ratios. Reread point number three from our abbreviated summary of Dalton's book again. Instead, he used his intellectual prowess to deduce that atoms of different elements such as carbon and hydrogen are not the same. That's because he really had no idea what an atom was like. Interestingly, Dalton made no attempt to actually describe the structure or composition of the atom itself. (Image from here.)ĭalton's concept of an atom was far more detailed than Democritus' and more importantly, it was based on experimental evidence. Two pages from John Dalton's 1808 book A New System of Chemical Philosophy. A chemical reactions never create or destroy atoms.When different element atoms get together in whole number rations, they form things called compounds.The atoms of one element must be different from the atoms of another element. They have the same size, mass, and chemical properties. When it comes to a single element, all of the atoms are identical.Elements are made of super small particles that we call atoms. 
In 1808, Dalton wrote a whole book about atomic theory called "A New System of Chemical Philosophy." Never fear as we have summarized the highlights of this piece of literature below. In fact, his work marks the beginning of the modern era of chemistry. (Image from here.)ĭalton's theories are considered special and monumental. 
He modernized the early definitions of matter and formulated a precise definition of the indivisible building blocks that we call atoms. He was an English scientist and school teacher living in the early 1800s. One guy who did not think Democritus was crazy was John Dalton. Democritus' work was not accepted by many of his contemporaries like Plato and Aristotle. We call these collective ideas the Atomic Theory. Democritus' atoms were also infinite in number, always moving and capable of joining together. To Democritus, atoms were small, hard particles that were all made of the same material but were different shapes and sizes. He named this smallest piece of matter atomos, meaning "not to cut." He reasoned that eventually the smallest piece would be obtained. C'mon you know you've tried it.ĭemocritus came to the conclusion that matter could not be divided into smaller and smaller pieces forever. He asked himself, "Self? Do you think matter can be divided into smaller and smaller pieces forever? Or is there a limit to the number of times a piece of matter can be divided?" It's kind of like the age old mystery of how many times can you fold a piece of paper. While most of us sit around thinking about pop music, celebrity gossip, or the latest zombie or vampire movie, Democritus focused his thoughts on more serious matters. This is crazy to think about, but over 2,400 years ago Democritus was sitting around Greece thinking about matter, which is anything that has mass and takes up volume. We heart this guy because he's the father of the Atomic Theory. 
Remember those early philosophers we talked about in the introduction? They're those snazzy thinkers whose curiosity about the world around them created the earliest ideas of science. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
